 ©Uday / Adobe Stock
©Uday / Adobe StockThe fusion process is the reaction that powers the sun. On the sun, in a series of nuclear reactions, four isotopes of hydrogen-1 are fused into a helium-4 with the release of a tremendous amount of energy.
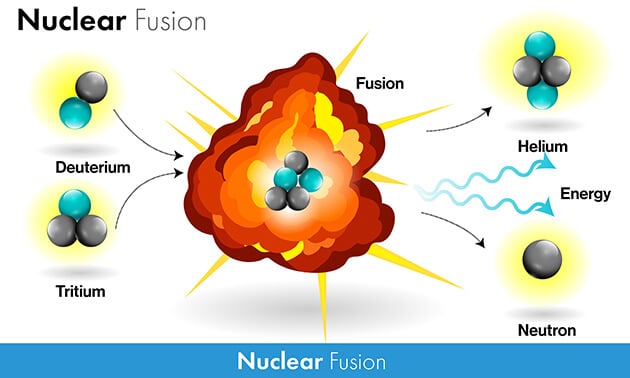
Here on earth, two other isotopes of hydrogen are used: H-2, called deuterium, and H-3, called tritium. Deuterium is a minor isotope of hydrogen, but it’s still relatively abundant. Tritium doesn’t occur naturally, but it can easily be produced by bombarding deuterium with a neutron.
The fusion reaction is shown in the following equation:
Major breakthrough in 2022
In December 2022, scientists at the U.S. National Ignition Facility in Livermore, California, achieved a major breakthrough in nuclear fusion research: energy gain. Energy gain, also known in this context as ignition, means that they were able to create a fusion reaction that released more energy than they put in by their lab's gigantic high-powered lasers.The scientists said they proved nuclear fusion can work on Earth, but we are still many years from being able to use it in power plants. Yet, scientists are optimistic that controlled fusion power will be achieved. The rewards are great — an unlimited source of clean energy.
Hydrogen bomb was first demonstration
The first demonstration of nuclear fusion — the hydrogen bomb — was conducted by the military. A hydrogen bomb is approximately 1,000 times as powerful as an ordinary atomic bomb.The isotopes of hydrogen needed for the hydrogen bomb fusion reaction were placed around an ordinary fission bomb. The explosion of the fission bomb released the energy needed to provide the activation energy (the energy necessary to initiate, or start, the reaction) for the fusion process.
Control issues with nuclear fusion
The goal of scientists for the last 50 years has been the controlled release of energy from a fusion reaction. If the energy from a fusion reaction can be released slowly, it can be used to produce electricity. It will provide an unlimited supply of energy that has no wastes to deal with or contaminants to harm the atmosphere — simply non-polluting helium.But achieving this goal requires overcoming three problems:
-
Temperature
-
Time
-
Containment
Temperature
The fusion process requires an extremely high activation energy. Heat is used to provide the energy, but it takes a lot of heat to start the reaction. Scientists estimate that the sample of hydrogen isotopes must be heated to approximately 40,000,000 K.K represents the Kelvin temperature scale. To get the Kelvin temperature, you add 273 to the Celsius temperature.
Now 40,000,000 K is hotter than the sun! At this temperature, the electrons have long since left the building; all that’s left is a positively charged plasma, bare nuclei heated to a tremendously high temperature. Presently, scientists are trying to heat samples to this high temperature through two ways — magnetic fields and lasers. Neither one has yet achieved the necessary temperature.Time
Time is the second problem scientists must overcome to achieve the controlled release of energy from fusion reactions. The charged nuclei must be held together close enough and long enough for the fusion reaction to start. Scientists estimate that the plasma needs to be held together at 40,000,000 K for about one second.Containment
Containment is the major problem facing fusion research. At 40,000,000 K, everything is a gas. The best ceramics developed for the space program would vaporize when exposed to this temperature.Because the plasma has a charge, magnetic fields can be used to contain it — like a magnetic bottle. But if the bottle leaks, the reaction won’t take place. And scientists have yet to create a magnetic field that won’t allow the plasma to leak.
Using lasers to zap the hydrogen isotope mixture and provide the necessary energy bypasses the containment problem. But scientists have not figured out how to protect the lasers themselves from the fusion reaction.

