When naming a branched alkane, you first need to locate and number the parent (longest) chain. You must then locate and identify all the substituents that stick off of the parent chain, including multiple and complex branches. Finally, you order the substituents alphabetically in front of the parent chain to construct the name of the branched alkane.
Identify and name multiple instances of the same constituent
What if the molecule contains more than one of the same substituent? For example, what if the compound has two methyl-group substituents on the molecule? In such a scenario, you don't name each of the methyl groups individually. Instead, you put a prefix in front of a single substituent name to indicate the number of these substituents that the molecule contains. (The following table contains a list of prefixes.)| Number of Identical Groups | Prefix |
|---|---|
| 2 | di |
| 3 | tri |
| 4 | tetra |
| 5 | penta |
| 6 | hexa |
| 7 | hepta |
| 8 | octa |
| 9 | nona |
| 10 | deca |
Students often forget that for every substituent you must explicitly list a number, even if the substituents are attached to the same carbon. If the molecule has three methyl groups coming off the parent chain, the substituent name is trimethyl, if it has four methyls, the name is tetramethyl, if it has five methyls, the name is pentamethyl, and so on.
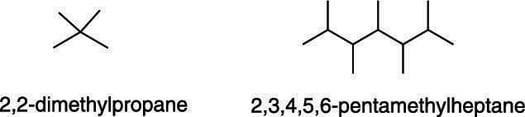
These prefixes are ignored when placing the substituents in alphabetical order (dimethyl would be alphabetized under m, for example). This figure shows two examples of molecules that have identical substituents. Note the use of commas to separate the numbers preceding the substituent name.
Identify and name complex substituents
Sometimes, substituents will branch and will have no common name, like the substituent shown here: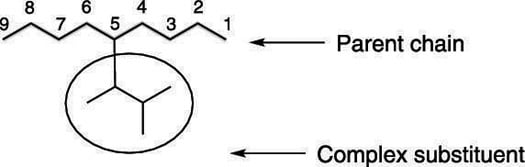
The main catch in naming complex substituents, however, is that the carbon that attaches the substituent to the parent chain must be the number-one carbon; thus, when you number the carbons in the substituent chain, you force the carbon that attaches to the main chain to be carbon number one. (The next figure shows an example of how this works.)
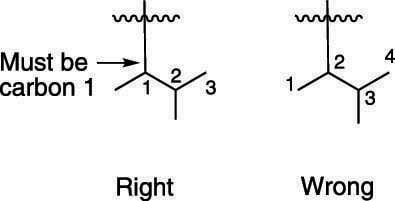
Because you're naming a substituent off the parent chain, the name must end with the suffix –yl, not –ane.
Now you can put all the parts of the name together. By convention, when a complex substituent is included in a name, the name of the complex substituent is set off with parentheses. So the name of the molecule in this example is 5-(1,2-dimethylpropyl)-nonane.
