Elimination reactions often compete with substitution reactions. As in substitution reactions, one possible mechanism for an elimination reaction is first-order elimination, or the E1 mechanism. The general form of an elimination reaction is shown in the first figure. In this reaction, a substrate (typically, an alkyl halide) eliminates one equivalent (unit) of acid to make an alkene.
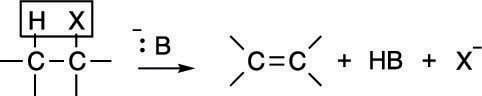
First-order elimination is somewhat less common than second-order (E2) elimination. The mechanism for the E1 reaction, like the mechanism for the SN1 reaction, has two steps and is shown in the next figure. First, the leaving group pops off to make the carbocation; this is the same first step as in the SN1 reaction. Then the base plucks off the hydrogen on an adjacent carbon to form the double bond.
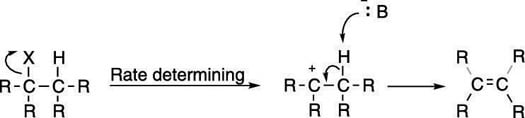
The first step of the mechanism — the formation of the carbocation — is the rate-determining step, and so the rate follows the equation: rate = k[substrate]. Because the base is not included in the rate equation, the strength of the base is unimportant to the rate of the reaction. E2 elimination is favored with strong bases, so you see E1 elimination typically only with weak bases.

