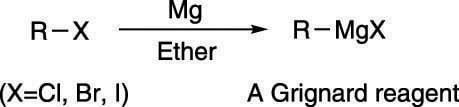
An extremely useful reaction for making alcohols is the Grignard reaction (pronounced grin-yard). To make alcohols using the Grignard reaction, you react a "Grignard reagent" with a carbonyl compound. Making a Grignard reagent is fairly simple: You simply add magnesium to an alkyl halide, as shown here, which inserts the magnesium into the C-X bond to make the Grignard reagent.
Making a Grignard reagent
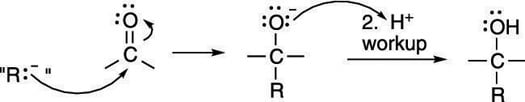
A Grignard reagent is an extremely powerful nucleophile (nucleus lover), and can react with electrophiles like carbonyl compounds. To determine the products made in a Grignard reaction, you can ignore the magnesium halide portion of the reagent (because this portion doesn't get involved in the reaction) and think of the Grignard reagent as acting as a carbanion (a negatively charged carbon atom) in disguise. The next figure illustrates this idea.
The mechanism for the addition of a Grignard reagent to a carbonyl is shown in the next figure.
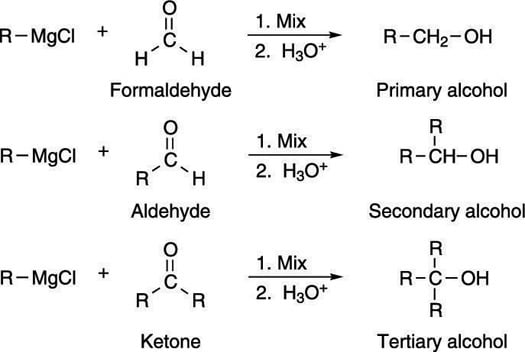
Although you can make only primary and secondary alcohols by reduction, you can make all kinds of alcohols using the Grignard reaction. If you react a Grignard reagent with formaldehyde, as shown in the next figure, you can make primary alcohols. If you react it with an aldehyde, you get secondary alcohols. If you react it with ketones, you get tertiary alcohols.
This figure shows the formation of alcohols via the addition of Grignard reagents to carbonyl compounds.