You will need to have a little chemistry facts under your sleeve for the ASVAB. Chemists study matter, and everything that has mass and takes up space — including your old Chevy that’s up on blocks and the mosquito buzzing around the room — is matter. All matter is made up of basic substances (building blocks) called elements.
Mass isn’t the same thing as weight. Weight has to do with the force that gravity exerts on mass. If you were in a gravity-free zone, you wouldn’t weigh anything, but you’d still be there, so you’d still exist and have mass.
Those mad scientists in the movies always seem to be chemists, but chemistry shouldn’t drive you crazy. Here’s a straightforward review of the chemistry you need to know for the General Science subtest.
Understanding the elements
The atom is the smallest part of an element that still retains the characteristics of that element. Every atom has particles — pieces of matter that are very, very small. Electrons are negatively charged particles that float around the atom’s nucleus, or core, which is made up of neutrons (particles with no charge) and protons (positively charged particles).
Each element has its own atomic number that’s equal to the number of protons. If an atom has one proton in its nucleus, it has the atomic number 1. Hydrogen is the only element with just one proton in its nucleus. Magnesium, which has 12 protons in its nucleus, is given the atomic number 12.
Atoms can combine with each other to form molecules. If those atoms are of two or more different elements, the molecule is called a compound. A compound can have very different properties from the elements that make it up. For example, table salt, which is mostly harmless, consists of two lethal elements — sodium and chlorine. But when combined, these elements make a compound that people ingest every day, salt.
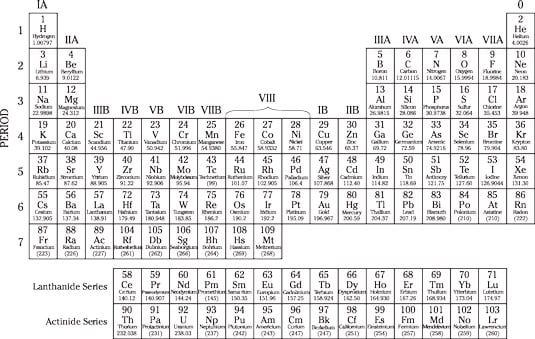
Sitting down at the periodic table
The periodic table (also known as the table of elements) classifies all elements, because scientists love to classify things. Elements are listed according to their atomic numbers (number of protons) and are arranged into families of similar elements.
The periodic table lists the atomic number, the abbreviation for each element, and its atomic weight, which is the average mass of one atom of the element. Looking at the periodic table, you can see that copper (Cu, atomic number 29) has an atomic weight of 63.546, which means that copper is much, much heavier than helium (He, atomic number 2), which has an atomic weight of 4.0026.
You don’t have to memorize these charts to do well on the ASVAB, but you should know the atomic numbers for common elements such as hydrogen (1), helium (2), carbon (6), nitrogen (7), oxygen (8), sodium (11), iron (26), copper (29), gold (79), mercury (80), lead (82), uranium (92), and plutonium (94).
Getting physical: Changing states
Particles of matter are always in motion. How much kinetic energy (motion energy) a particle has determines whether the matter is a solid, liquid, or gas in its normal state. Gas particles move around very quickly, liquid particles move more slowly, and solid particles move much more slowly than either of the other two.
When heat or cold is applied to matter, the kinetic energy of the matter changes; therefore, the nature of the substance can change. Heat applied to water changes the water from a liquid to a gas (steam), and cold applied to water changes it from a liquid to a solid (ice). When physical changes occur, the molecule itself remains the same. For example, water is still made of hydrogen and oxygen, no matter which state it’s in.
Causing a chemical reaction
Unlike physical changes, chemical reactions create new molecules. For example, when iron rusts, a chemical change occurs. The rust isn’t the same molecule as the iron.
In a chemical reaction, two kinds of substances are present:
Reactants: The elements or molecules involved in the reaction
Products: The elements or molecules that result from the chemical reaction

