Alkenes, which are molecules containing carbon-carbon double bonds, have the possibility of having stereoisomers, just as ring systems do. This is because, unlike carbon-carbon single bonds, which are free to rotate, double bonds are fixed and rigid. In other words, rotation around carbon-carbon double bonds is not possible at reasonable temperatures.
Molecules that have the same connectivity of atoms, but different orientations of those atoms in space are called stereoisomers.
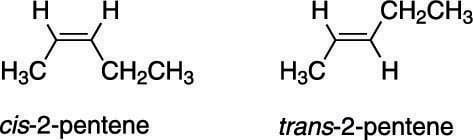
Consider the case of 2-pentene, shown here. In this alkene, two stereoisomers are possible. One stereoisomer, called the cis stereoisomer, has both of the double-bond hydrogens on the same side of the double bond, while the other stereoisomer, called the trans stereoisomer, has the two hydrogens on opposite sides of the double bond. In general, when two identical groups are on the same side of the double bond, the molecule is said to possess cis stereochemistry; when two identical groups are on opposite sides of the double bond, the molecule is said to possess trans stereochemistry.
You can only have cis-trans stereochemistry in rings and on double bonds. You can't have cis-trans isomers on single bonds due to the rapid free rotation of these bonds at room temperature.

