Alkyne reactions are similar to those of the alkenes, and these reactions use reagents similar to those used in alkene reactions. Alkynes can form a variety of functional groups, including tetrabromides, alkanes, alkenes, ketones, and aldehydes.
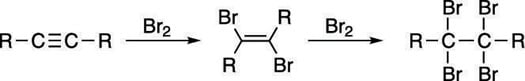
Brominate an alkyne to form a tetrabromide
Bromine reacts with the pi bond in alkynes to make the dibromide via the same mechanism as bromine addition to alkenes (think "bromonium ion"). Because there are two pi bonds in an alkyne, two equivalents (units) of bromine can be added to make a tetrabromide, as shown here.
Saturate an alkyne to form an alkane
Alkynes can also be reduced to alkanes by bubbling two equivalents of hydrogen gas (H2) over the alkyne in the presence of a metal catalyst as shown in the next figure. This catalyst is usually palladium on carbon (Pd/C), but platinum (Pt) is also sometimes used.
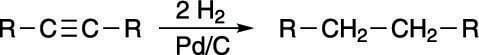
Add one hydrogen molecule to form an alkene
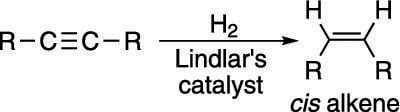
Stopping the reaction of alkynes with hydrogen at the alkene stage is possible because alkenes are somewhat less reactive than alkynes, but this reaction requires a special catalyst. To reduce an alkyne to the cis alkene, you use Lindlar's catalyst, which is a cocktail of palladium (Pd) powder made less reactive with added lead (Pb) and quinoline (C9H7N). (In reaction diagrams, instead of writing out all the components of the catalyst, chemists often write "Lindlar's catalyst" over or under the arrow.) Lindlar's catalyst is not as reactive as palladium on carbon (Pd/C) and generates the cis alkene, as shown here.
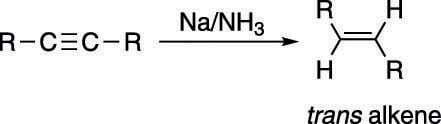
To convert an alkyne to the trans alkene, you use sodium metal (Na) in liquid ammonia (NH3), as shown in the next figure.
Oxymercurate an alkyne to form a ketone
Reacting alkynes with mercury, water, and acid, you may expect the reaction to make the alcohol on the double bond, just as mercuric acetate reacts with alkenes to make the Markovnikov alcohol. Indeed, the Markovnikov enol (an alcohol on a double bond) is formed in which the alcohol group is placed on the more-substituted carbon. However, enols are unstable and rapidly convert into ketones, as shown here.
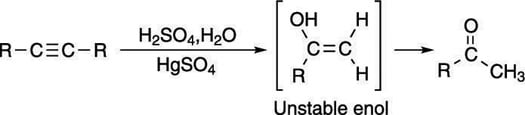
The reaction that converts the enol into the ketone is called a tautomerization reaction, and both the enol and the ketone are considered tautomers of each other. Tautomers are molecules that differ only in the placement of a double bond and a hydrogen.
Apply a hydroboration reaction to an alkyne to form an aldehyde
The hydroboration reaction of alkynes works in the same way as the hydroboration reaction of alkenes, and forms the anti-Markovnikov product. In the case of the hydroboration of alkynes, the product is an enol with the alcohol group on the least-substituted carbon, as shown in the next figure. As with the oxymercuration reaction, this enol is unstable and tautomerizes to the aldehyde.
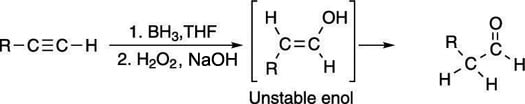
Oxymercuration and hydroboration of carbon-carbon triple bonds are useful primarily with terminal alkynes because with terminal alkynes, you get only a single product (with hydroboration, you get the aldehyde; with oxymercuration, you get the ketone). With internal alkynes, both sides of the alkyne are equally substituted, so water can be added equally well to either side of the triple bond, so these reactions yield mixtures of two products.

