Combustion reactions of fuels and foods are types of redox reactions that are essential for life and civilization — because heat is the most important product of these reactions.
Following are common examples of combustion reactions:
The burning of coal, wood, natural gas, and petroleum heats our homes and provides the majority of our electricity.
The combustion of gasoline, jet fuel, and diesel fuel powers our transportation systems.
The combustion of food powers our bodies.
Have you ever wondered how the energy content of a fuel or food is measured? An instrument called a bomb calorimeter is used to measure energy content. The following diagram shows the major components of a bomb calorimeter.
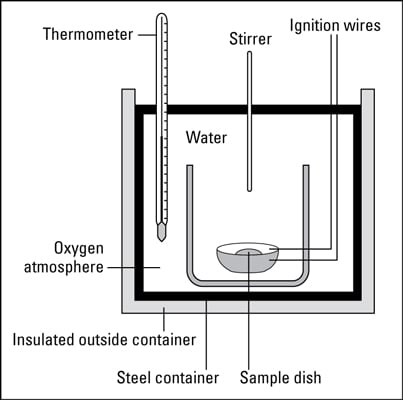
To measure the energy content of fuels, a known mass of the material to be measured is placed into a sample cup and sealed. The air is removed from the sample cup and replaced with pure oxygen. The cup is then placed in the calorimeter with a known amount of water covering it.
The initial temperature of the water is measured, and then the sample is ignited electrically. The rise in the temperature is measured, and the number of calories of energy that is released is calculated.
A calorie is the amount of energy needed to raise the temperature of 1 gram of water 1 degree Celsius. The complete combustion of a large kitchen match, for example, gives you about one kilocalorie of heat.
The caloric content of foods can be determined in exactly the same fashion. Chemists report the results in calories or kilocalories, while nutritionists report the results in nutritional Calories.
A nutritional Calorie is equal to a chemist’s kilocalorie (1,000 calories). A 300 Calorie candy bar produces 300,000 calories of energy. Unfortunately, not all that energy is required immediately, so some is stored in compounds such as fats.

