Here is a brief exploration of the Standard Model of particle physics and how it relates to string theory. Any complete string theory will have to include the features of the Standard Model and also extend beyond it to include gravity as well.
Today scientists know that these atoms are not, as the Greeks imagined, the smallest chunks of matter. Scientists quickly realized that atoms had multiple parts inside of them:
Negatively charged electrons circling the nucleus
Positively charged nucleus
The particles that compose the nucleus (it’s made up of smaller pieces, too) and electrons are among the particles, along with several others, that the Standard Model of particle physics explains, and ultimately that string theory should also explain.
Discovering the electron
The electron is a negatively charged particle contained within the atom. It was discovered in 1897 by British physicist J.J. Thomson, though charged particles (including the name “electron”) had been hypothesized earlier.
Some physicists had already hypothesized that units of charge might be flowing around in electrical apparatus. (Benjamin Franklin proposed such an idea as early as the 1700s.) Technology only caught up to this idea in the late 1800s, with the creation of the cathode ray tube, shown in this figure.
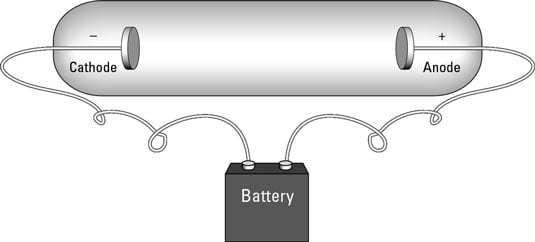
In a cathode ray tube, a pair of metal disks is connected to a battery. The metal disks are placed inside a sealed glass tube that contains no air — a vacuum tube. The electrical voltage causes one of the metal plates to become positively charged (an anode) and one to become negatively charged (the cathode, from which the device gets its name). Cathode ray tubes are the basis of traditional television and computer monitor tubes.
When the electrical current was switched on, the tube would begin to glow green. In 1897, Thomson was head of the Cavendish laboratory in Cambridge, England, and set about to test the properties of this cathode ray tube glow. He discovered that the glow was due to a beam of negatively charged particles flying between the plates.
These negatively charged particles later came to be called electrons. Thomson also figured out that the electrons were incredibly light — 2,000 times lighter than a hydrogen atom.
Thomson not only discovered the electron, but he theorized that the electron was part of the atom (atoms weren’t a completely accepted idea at the time) that somehow got knocked free from the cathode and flowed through the vacuum to the anode. With this discovery, scientists began discovering ways to explore the inside of atoms.
The nucleus is the thing in the middle
In the center of the atom is a dense ball of matter, called a nucleus, with a positive electrical charge. Shortly after electrons were discovered, it became clear that if you extracted an electron from an atom, the atom was left with a slightly positive electrical charge.
For a while, the assumption was that the atom was a positively charged mass that contained negative electrons inside of it, like pieces of negatively charged fruit in a positively charged fruitcake. The entire fruitcake would be neutral unless you extracted some fruit from it.
In 1909, however, an experiment by Hans Geiger and Ernest Marsden, working under Ernest Rutherford, challenged this picture. These scientists fired positively charged particles at a thin sheet of gold foil. Most of the particles passed straight through the foil, but every once in a while one of them bounced back sharply.
Rutherford concluded that the positive charge of the gold atom wasn’t spread throughout the atom in the fruitcake model, but was concentrated in a small positively charged nucleus, and that the rest of the atom was empty space. The particles that bounced were the ones that hit this nucleus.

