Alcohols and amines are fairly easy to identify in the IR spectrum, based on their relative locations and shapes. The first thing you'll notice is that both of these functional groups appear to the left of the C-H absorptions, which always occur between 2,800 cm–1 to 3,000 cm–1 in the IR spectrum.
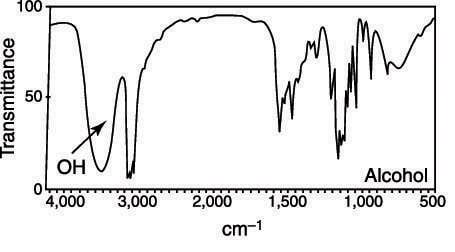
In addition to appearing to the left of the C-H absorptions, alcohols are also very broad, fat absorptions, as shown in the figure. Because of their broadness, they're very easy to spot.
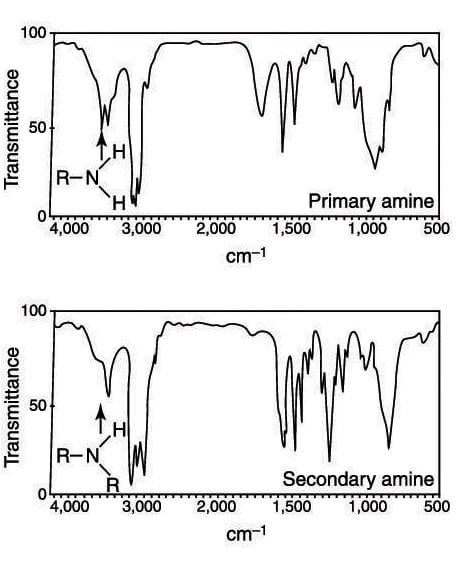
Primary amines (amines substituted by only one R group and abbreviated RNH2) are also very easy to spot. They appear to the left of the C-H absorptions in the spectrum in about the same region as alcohol absorptions. Primary amines consist of two small peaks, and the overall effect is to make the absorptions of a primary amine look like a cow udder, as shown in the second figure). Secondary amines (amines substituted by two R groups and abbreviated R2NH) consist of only a single absorption in that region.
A common mistake is to confuse secondary amine absorption with alcohol absorptions, because they both occur in the same region of the spectrum. Usually, secondary amine absorptions are somewhat thinner and sharper than the broad and rounded absorptions produced by alcohols. Only practice with problems dealing with IR spectra will make you confident in telling the difference between amine and alcohol absorptions.

