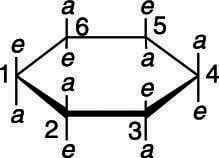
For example, if you were asked to draw the most stable conformation of cis-1,3-dimethylcyclohexane, both substituents could be on the top of the ring or both could be on the bottom of the ring, as shown here. (Recall that cis means that both substituents are on the same side of the ring.) As the figure shows, to get the cis stereochemistry, either both of the substituents could be equatorial (both e) or both could be axial (both a).
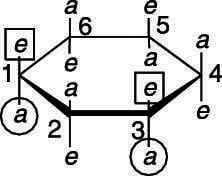
Putting large groups in the equatorial position to eliminate 1,3-diaxial strain is energetically favorable.
Because large groups prefer to be equatorial, the most stable conformer for cis-1,3-dimethylcyclohexane is the diequatorial conformer, shown here. The diaxial conformer would be higher in energy.
If cyclohexane has two substituents and one has to be placed axial and one equatorial (as is the case in trans-1,2-disubstituted cyclohexanes), the lowest-energy conformation will be the one in which the bigger group goes in the equatorial position and the smaller group goes in the axial position.

