After you number the parent chain, if you want to name the branched alkane, you need to determine the names of all the substituents that stick off of the parent chain, and then order the substituents alphabetically in front of the parent chain.
This figure shows an example of a branching alkane. As you can see, the alkane with the formula C4H10 can have two possible chemical structures, or isomers — one alkane that’s a straight chain (butane), and one alkane that’s branched (isobutane).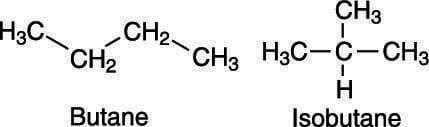
The isomers of C4H10
Find the longest chain in a branched alkane
The first, and potentially the trickiest, step in naming a branched alkane is to find the longest chain of carbon atoms in the molecule. This task can be tricky because, as a reader of English, you’re so used to reading from left to right.Often, however, the longest chain of carbons is not the chain that follows simply from left to right, but one that snakes around the molecule in different directions. Organic professors like to make the parent chain one that curves around the molecule and doesn’t necessarily flow from left to right, so you have to keep on your toes to make sure that you’ve spotted the longest carbon chain.
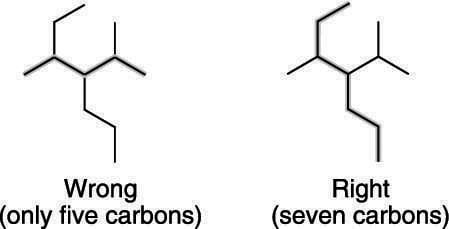
The wrong and right ways to count the parent chain
Number the longest chain in a branched alkane
Number the parent chain starting with the end that reaches a substituent first. A chain can always be numbered in two ways. For the molecule in this example, the numbering could start at the top and go down, or it could start at the bottom and go up. The correct way to number the parent chain is to start with the end that reaches the first substituent sooner. A substituent is organic-speak for a fragment that comes off of the parent chain.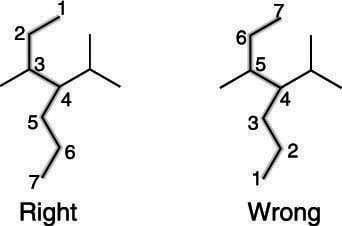
The right and wrong ways to number the carbons of the parent chain

