One double bond, one degree of unsaturation
The degrees of unsaturation in a molecule are additive — a molecule with one double bond has one degree of unsaturation, a molecule with two double bonds has two degrees of unsaturation, and so forth.Just as the formation of a double bond causes two hydrogens to be lost, the formation of a ring also results in the loss of two hydrogens, so every ring in the molecule also adds one degree of unsaturation.
For every triple bond, two degrees of unsaturation are added to a molecule, because a molecule must lose four hydrogens to make a triple bond. Some examples of three-carbon molecules with different numbers of degrees of unsaturation are shown here.
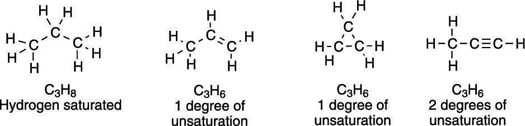
The goal: Using a molecular formula
More important than determining the number of degrees of unsaturation from a molecular structure is being able to determine the number of degrees of unsaturation from a molecular formula. The number of degrees of unsaturation can be determined from the molecular formula using the following equation.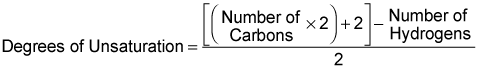
With this equation, the number of degrees of unsaturation can be determined for any hydrocarbon whose molecular formula is known. (For compounds whose structure and formula are not known, chemists use an instrumental technique called mass spectrometry to determine the molecular formula of the compound.)
But what about molecules that contain atoms other than hydrogen and carbon? In such cases, you need to convert these multi-atom molecular formulas into equivalent formulas that contain just carbon and hydrogen so they can be plugged into the preceding equation. To do so, you use the following conversion factors:
-
Halogens (F, Cl, Br, I): Add one hydrogen to the molecular formula for each halogen present.
-
Nitrogen: Subtract one hydrogen for each nitrogen present.
-
Oxygen or sulfur: Ignore.

