Hydration, or adding water across a double bond to make an alcohol, is a reaction that's similar to the addition of a hydrohalic acid across a double bond. Two different reactions accomplish the hydration. The first reaction adds the alcohol (OH group) to the most substituted carbon on the double bond to make the Markovnikov product, and the complementary reaction puts the alcohol on the least substituted carbon in the double bond to make the anti-Markovnikov product.
Convert alkenes using Markovnikov addition
To make the Markovnikov product where the alcohol adds to the most substituted carbon, you react the alkene with mercuric acetate, Hg(OAc)2 and water, followed by addition of sodium borohydride, NaBH4, as shown here.
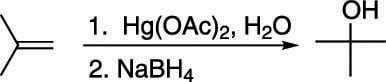
The numbers over (or under) the reaction arrow indicate separate steps. In the case of oxymercuration-demercuration, the numbers specify that mercuric acetate is added first, followed by sodium borohydride. When no numbers are present over (or under) the arrow, this indicates that reagents are all added together in the same pot.
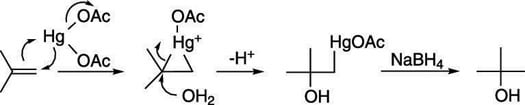
The mechanism for the oxymercuration-demercuration involves an attack of the double bond on the mercuric acetate to make a three-membered ring intermediate (called a mercurinium ion), as shown in the next figure. Water then attacks the most highly substituted carbon to make the mercurial alcohol (after the loss of a proton). In the second step (when NaBH4 is added), sodium borohydride replaces the mercuric portion with hydrogen.
Convert alkenes using anti-Markovnikov addition
With oxymercuration-demercuration, you have a reaction that converts alkenes into Markovnikov-product alcohols. To make the alcohol on the least-substituted carbon (called the anti-Markovnikov product) you use hydroboration, as shown in the next figure. The addition of borane (BH3) in tetrahydrofuran solvent (THF) to the alkene, followed by the addition of hydrogen peroxide (H2O2) and sodium hydroxide (NaOH), make the anti-Markovnikov alcohol.
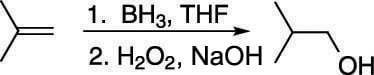
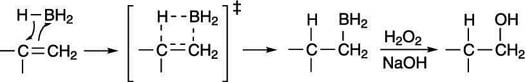
The mechanism for hydroboration involves the cyclic transition state shown in the next figure. Borane adds to the least substituted side of the double bond to make the alkyl borane. Because the addition is concerted (both the hydrogen and BH2 are added simultaneously), the borane and hydrogen must add to the same face of the carbon-carbon bond (two groups adding to the same face of a double bond is called syn addition). In the second step, hydrogen peroxide (H2O2) in the presence of sodium hydroxide (NaOH) substitutes a hydroxyl group (OH) for the boryl unit (BH2) to make the anti-Markovnikov alcohol.