Chemists are finicky, tinkering types. They usually want to change reaction rates to suit their own needs. What can affect rates, and why? Temperature, concentration, and catalysts influence rates as follows:
Reaction rates tend to increase with temperature. This trend results from the fact that reactants must collide with one another to have the chance to react. If reactants collide with the right orientation and with enough energy, the reaction can occur. So the greater the number of collisions and the greater the energy of those collisions, the more actual reacting takes place. An increase in temperature corresponds to an increase in the average kinetic energy of the particles in a reacting mixture — the particles move faster, colliding more frequently and with greater energy.
Increasing concentration tends to increase the reaction rate. The reason for this trend also has to do with collisions. A higher concentration means that more reactant particles are closer together, so they undergo more collisions and have a greater chance of reacting. Increasing the concentration of reactants may mean dissolving more of those reactants in solution.
Some reactants aren't completely dissolved but come in larger, undissolved particles. In these cases, smaller particles lead to faster reactions. Smaller particles expose more surface area, making a greater portion of the particle available for reaction.
Catalysts increase reaction rates. Catalysts don't themselves become chemically changed, and they don't alter the amount of product a reaction can eventually produce (the yield). An example from early childhood fits here. When you're learning to ride a bike, you might get a push from your parents to help you get going. However, after that push, the pedaling is entirely up to you. Your top speed and end destination are still entirely regulated by your ability to pedal and steer the bike, but that push (the catalyst) helped you get up to speed more quickly.
Catalysts can operate in many different ways, but all those ways have to do with decreasing activation energy, the energetic hill reactants must climb to reach a transition state, the highest-energy state along a reaction pathway. Lower activation energies mean faster reactions. The figure shows a reaction progress diagram, the energetic pathway that reactants must traverse to become products.
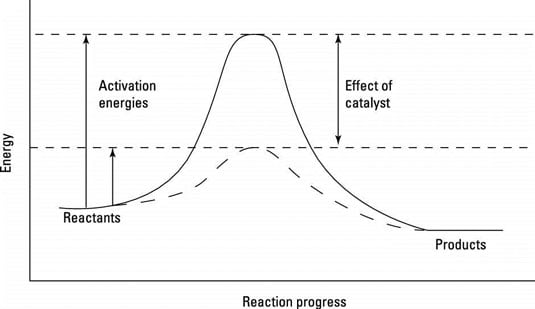 A reaction progress diagram.
A reaction progress diagram.
As an example, consider the following reaction:
If 1 mol of H2 reacts with 1 mol of Cl2 to form 2 mol of HCl, does the reaction occur more rapidly in a 5 L vessel or a 10 L vessel? Does it occur more rapidly at 273 K or 293 K? Why?
The reaction occurs more rapidly in the 5 L vessel because the concentration of reactant molecules is higher when they occupy the smaller volume. At higher concentrations, more collisions occur between reactant molecules. At higher temperatures, particles move with greater energy, which also produces more collisions and collisions of greater force. So the reaction occurs more rapidly at 293 K.

