In a hydrogen atom, the wave functions change as you change the orbital radius, r. So what do the hydrogen wave functions look like? Given that
looks like this:
Here are some other hydrogen wave functions:
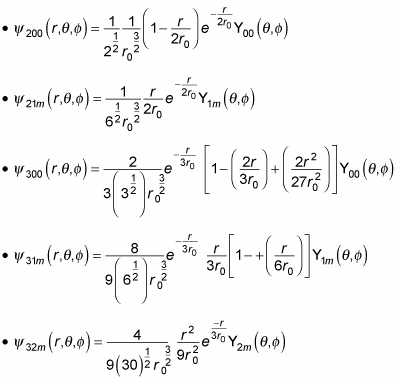
Note that
behaves like rl for small r and therefore goes to zero. And for large r,
decays exponentially to zero. So you’ve solved the problem of the wave function diverging as r becomes large — and all because of the quantization condition, which cut the expression for f(r) from an exponent to a polynomial of limited order. Not bad.
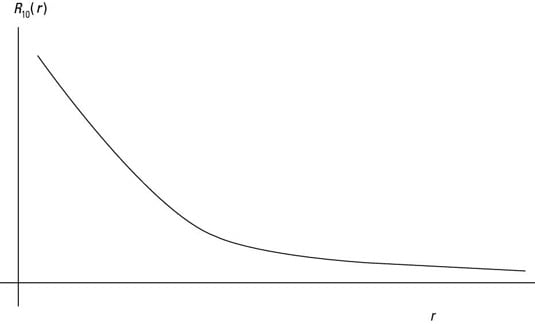
The radial wave function R10(r).
You can see the radial wave function R10(r) in the first figure. R20(r) appears in the second figure. And you can see R21(r) in the last figure.

R20(r).

R21(r).

