In physics, a substance’s specific gravity is the ratio of that substance’s density to the density of water at 4 degrees Celsius. Because the density of water at 4 degrees Celsius is 1,000 kg/m3, that ratio is easy to find. For example, the density of gold is 19,300 kg/m3, so its specific gravity is the following:
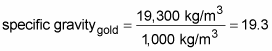
Specific gravity has no units because it’s a ratio of density divided by density, so all units cancel out. Therefore, the specific gravity of gold is simply 19.3.
Anything with a density greater than 1,000 sinks in pure water at 4 degrees Celsius, and anything with a density less than 1,000 floats. As you’d expect, gold, with a density of 19,300 kg/m3, sinks. Ice, on the other hand, with a density of 917 kg/m3, floats.
So how can a ship, which is made of metal with a specific gravity very much greater than water, float? The ship floats because of the shape of its hull. The ship is mostly hollow and displaces water weighing more than the weight of the ship. Averaged throughout, the ship is less dense than the water overall, so the effective specific gravity of the ship is less than that of water.

