The debate over nuclear power plants has been going on for some time, with nuclear physicists and lawmakers alike throwing around terms like nuclear fission, critical mass, and chain reaction. But how does nuclear fission work, exactly?
In the 1930s, scientists discovered that some nuclear reactions can be initiated and controlled. Scientists usually accomplished this task by bombarding a large isotope with a second, smaller one — commonly a neutron. The collision caused the larger isotope to break apart into two or more elements, which is called nuclear fission. Figure 1 shows the equation for the nuclear fission of uranium-235.
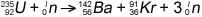
Figure 1: The equation for nuclear fission.
Reactions of this type also release a lot of energy. Where does the energy come from? Well, if you make very accurate measurement of the masses of all the atoms and subatomic particles you start with and all the atoms and subatomic particles you end up with, and then compare the two, you find that there's some "missing" mass. Matter disappears during the nuclear reaction. This loss of matter is called the mass defect. The missing matter is converted into energy.
You can actually calculate the amount of energy produced during a nuclear reaction with a fairly simple equation developed by Einstein: E = mc2. In this equation, E is the amount of energy produced, m is the "missing" mass, or the mass defect, and c is the speed of light, which is a rather large number. The speed of light is squared, making that part of the equation a very large number that, even when multiplied by a small amount of mass, yields a large amount of energy.
Take another look at the equation for the fission of U-235. Notice that one neutron was used, but three were produced. These three neutrons, if they encounter other U-235 atoms, can initiate other fissions, producing even more neutrons. It's the old domino effect. In terms of nuclear chemistry, it's a continuing cascade of nuclear fissions called a chain reaction. The chain reaction of U-235 is shown in Figure 2.
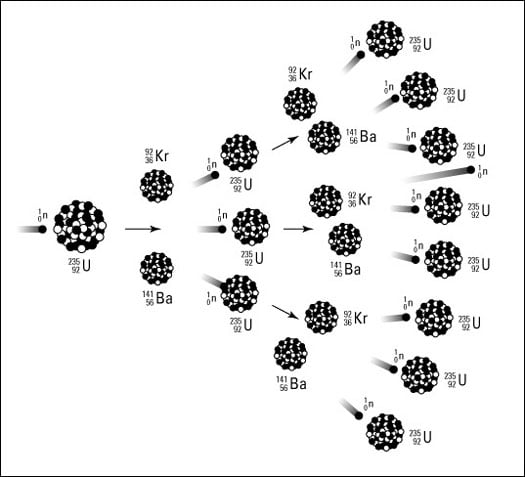
Figure 2: Chain reaction.
This chain reaction depends on the release of more neutrons than were used during the nuclear reaction. If you were to write the equation for the nuclear fission of U-238, the more abundant isotope of uranium, you'd use one neutron and only get one back out. You can't have a chain reaction with U-238. But isotopes that produce an excess of neutrons in their fission support a chain reaction. This type of isotope is said to be fissionable, and there are only two main fissionable isotopes used during nuclear reactions — uranium-235 and plutonium-239.
A certain minimum amount of fissionable matter is needed to support a self-sustaining chain reaction, and it's related to those neutrons. If the sample is small, then the neutrons are likely to shoot out of the sample before hitting a U-235 nucleus. If they don't hit a U-235 nucleus, no extra electrons and no energy are released. The reaction just fizzles. The minimum amount of fissionable material needed to ensure that a chain reaction occurs is called the critical mass. Anything less than this amount is called subcritical.

